 
This ability of binding 4 different atoms for a 3-D structure is unique to organic compounds. Video 2 – sp3 Hybridization and Bond AnglesĬarbon must have 4 bonds. Click here for an in-depth overview (+ videos) of sp³, sp² and sp Hybridization, Geometry and Bond Angles. These are crucial to understanding reactions. Learning these concepts will help you recognize strong/weak bonds and bonds that can or can’t be broken. Video 1 – Ionic, Polar Covalent and Non-Polar Covalent Bonding Have trouble classifying primary, secondary, and tertiary carbon atoms? You can't get far without this skill, and this trick makes its simple! I'll also cover molecules containing oxygen bonds, nitrogen, and more. Pre-Basics Video – Skeletal StructuresĬonfused by those sticks and lines masquerading as molecules? In this video, you’ll learn how to identify molecules when written in line structure. Worried you're behind? Go back to the ‘Gen Chem Review for Orgo' series first and test your knowledge with the Gen Chem for Orgo Quiz!įor a quick review, stop and watch my ‘ General Chemistry Review for Organic Chemistry Students‘ video. That's why this video series is designed to teach you these very foundation topics that were touched on in general chemistry. Write the Lewis structure and chemical formula of the compound with a molar mass of about 70 g/mol that contains 19.7% nitrogen and 80.3% fluorine by mass, and determine the formal charge of the atoms in this compound.Success in organic chemistry requires a strong foundation and understanding of the basics.Write Lewis structures for each of the four compounds and determine the formal charge of the iodine atom in each molecule: Iodine forms a series of fluorides (listed below). 
Is the actual structure consistent with the formal charges? 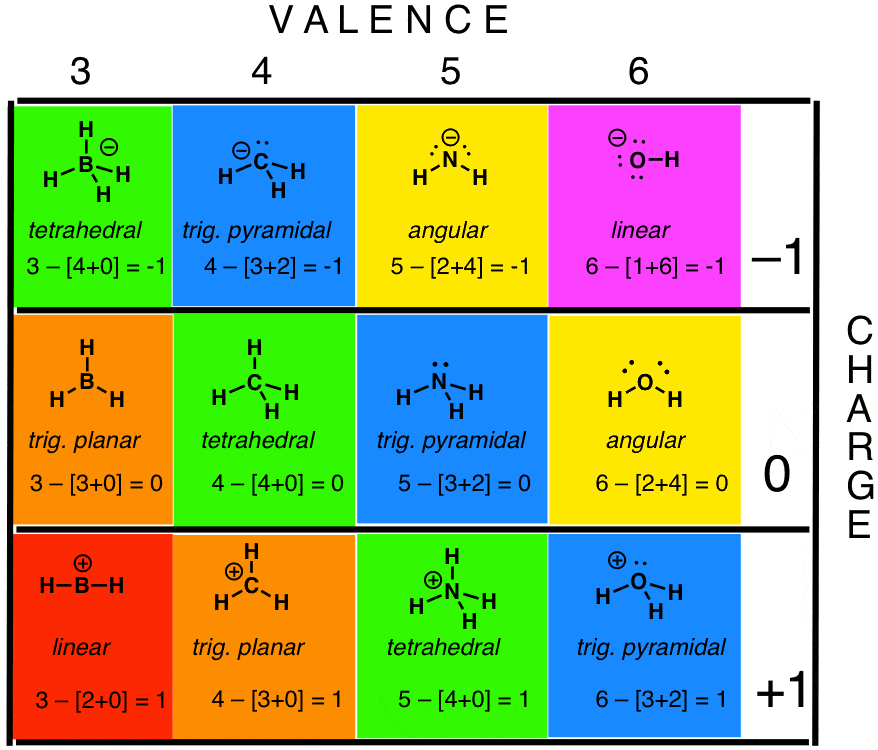
Draw the structure of hydroxylamine, H 3NO, and assign formal charges look up the structure.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in sulfur dioxide: OSO or SOO?.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in hypochlorous acid: HOCl or OClH?.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in nitrosyl chloride: ClNO or ClON?.Determine the formal charge on each atom in each of the resonance structures: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
